Fed Circuit Watch: Broad Wins Latest CRISPR Court Battle
On September 10, 2018, the Court of Appeals for the Federal Circuit decided Regents of the Univ. of Calif. v. Broad Inst., Inc.,1 in the latest court battle in the CRISPR patent challenge pitting three of the nation’s largest research universities against each other.
CRISPR, or “Clustered Regularly Interspaced Short Palindromic Repeats,” is a family of palindromic DNA sequences in prokaryotic cellular structures, like bacteria and archaea, and is more commonly associated with the novel gene editing (aka, genome editing) technology. Although CRISPR is a naturally-occurring acquired immunity system in prokaryotes, the cutting-edge gene editing research focused on how to modify these genetic codes in order to correct genetic abnormalities2 or to fight against certain viruses (e.g., Vlachakis).3 Certain associated proteins recognize and cut these foreign DNA/RNA from the strands. One of these proteins, Cas9, has been the subject of a lot of research at universities and institutes around the world. At labs at U.C. Berkeley and the University of Vienna (W.U.) (collectively “U.C.”), Drs. Jennifer Doudna and Emmanuelle Charpentier, respectively, studied the CRISPR system of the S. pyogenes bacterium, which relies on the Cas9 protein. They discovered that the Cas9 endonuclease was composed of four components, including two RNA molecules: crRNA and tracrRNA. The pair modified these RNA into a single RNA which could find and cut DNA target sequences.4 This method was what became the basis for several patents filed with Drs. Doudna and Charpentier as inventors, and the U.C. and W.U. as assignees. On the East Coast, Drs. Feng Zhang and George Church of MIT/Broad Institute and Harvard University (collectively “Broad”), respectively, focused on gene editing using CRISPR/Cas9 in human cell cultures. Their research then extended to eukaryotes. This became the basis for a family of patents and applications with Drs. Zhang and Church as inventors, and MIT, Broad, and Harvard as assignees.
This then leads up to the case at-hand.
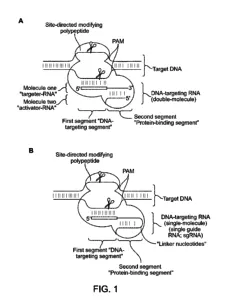 Source: U.S. Patent Application Publication No. 2014/0068797 A1, Mar. 6, 2014, to Jennifer A. Doudna, Martin Jinek, Emmanuelle Charpentier, James Harrison Doudna Cate, Wendell Lim, Lei Qi (inventors); University of Vienna & Regents of the University of California (assignees)
Source: U.S. Patent Application Publication No. 2014/0068797 A1, Mar. 6, 2014, to Jennifer A. Doudna, Martin Jinek, Emmanuelle Charpentier, James Harrison Doudna Cate, Wendell Lim, Lei Qi (inventors); University of Vienna & Regents of the University of California (assignees)
The U.C.’s patent application, Ser. No. 13/842,859 (‘859), published as Publication No. 2014/0068797, was filed on March 15, 2013, with a priority date of May 25, 2012. It was still pending with the USPTO as of September 14, 2018. Broad’s patent application was filed October 15, 2013, which later matured as U.S. Patent No. 8,697,359 (‘359), issued on April 15, 2014. It has a priority date of December 12, 2012. Because both applications were filed before March 16, 2013, both the ‘859 application and the ‘359 patent were governed by pre-AIA rules. As such, on January 11, 2016, the USPTO, while the ‘859 application was undergoing examination, declared an interference under 35 U.S.C. §135(a) and referred the case to the PTAB (MPEP 2301), subject to 35 U.S.C. §102(g) (pre-AIA).
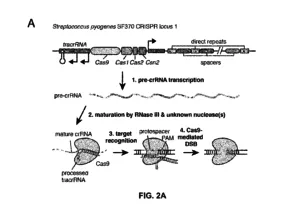 Source: U.S. Patent No. 8,697,359 B1, Apr. 15, 2014, to Feng Zhang (inventor); Broad Institute Inc., Massachusetts Institute of Technology (assignees)
Source: U.S. Patent No. 8,697,359 B1, Apr. 15, 2014, to Feng Zhang (inventor); Broad Institute Inc., Massachusetts Institute of Technology (assignees)
Two claims from each of the respective parties’ application or patent were deemed representative: claim 165 of the ‘859 application:
- A method of cleaving a nucleic acid comprising
contacting a target DNA molecule having a target sequence with an engineered and/or non-naturally-occurring Type II Clustered Regularly Interspaced Short Palindromic Repeats (CRISPR) – CRISPR associated (Cas) (CRISPR-Cas) system comprising
a) a Cas9 protein; and
b) a single molecule DNA-targeting RNA comprising
i) a targeter-RNA that hybridizes with the target sequence, and
ii) an activator-RNA that hybridizes with the targeter-RNA to form a double-stranded RNA duplex of a protein-binding segment,
wherein the activator-RNA and the targeter-RNA are covalently linked to one another with intervening nucleotides,
wherein the single molecule DNA-targeting RNA forms a complex with the Cas9 protein,
whereby the single molecule DNA-targeting RNA targets the target sequence, and the Cas9 protein cleaves the target DNA molecule.
And claim 1 of the ‘359 patent:
- A method of altering express of at least one gene product comprising introducing into a eukaryotic cell containing and expressing a DNA molecule having a target sequence and encoding the gene product an engineered, non-naturally occurring Clustered Regularly Interspaced Short Palindromic Repeats (CRISPR) – CRISPR associated (Cas) (CRISPR-Cas) system comprising one or more vectors comprising:
a) a first regulatory element operable in a eukaryotic cell operably linked to at least one nucleotide sequence encoding a CRISPR-Cas system guide RNA that hybridizes with the target sequence, and
b) a second regulatory element operable in a eukaryotic cell operably linked to a nucleotide sequence encoding a Type-II Cas9 protein,
wherein components (a) and (b) are located on same or different vectors of the system, whereby the guide RNA targets the target sequence and the Cas9 protein cleaves the DNA molecule, whereby expression of the at least one gene product is altered; and, wherein the Cas9 protein and the guide RNA do not naturally occur together.
As it can be seen, the U.C. claim is very broadly worded to cover the entire process of Cas9 RNA-targeting of the DNA target to be cut, while the Broad claims are focused on CRISPR-Cas9 in eukaryotic cells.
Because §102(g) requires the USPTO to issue a patent to the first-to-invent inventor where two parties claim patentably indistinct subject matter, the U.C. would procedurally have had the upper hand in the proceeding. Broad then moved to terminate the interference, arguing that its claims were patentably distinct from the U.C. claims because a person of ordinary skill in the art (“POSITA”) would not have had a reasonable expectation of success that the CRISPR-Cas9 system would work in eukaryotic cells. The PTAB agreed, and found no interference-in-fact. The U.C. appealed.
The Fed Circuit panel, composed of Chief Judge Prost, and Judges Schall and Moore, with Judge Moore writing for a unanimous court, noted that an interference-in-fact exists where one parties’ claims renders obvious the subject matter of the other parties’ claims. Further, the appropriate standard of review is the substantial evidence standard. Judge Moore turned to analyze the two PTAB rationales in finding no interference-in-fact. She focused on both the extrinsic evidence – specifically, expert witness testimony – and the intrinsic evidence. First, Broad’s expert testified that there were concerns involving the CRISPR-Cas9 system that a POSITA would not have a reasonable expectation of success in eukaryotic cells. This was supported by the U.C.’s own expert who testified there were issues with applying CRISPR-Cas9 in eukaryotic cells, which included toxicity development in the nucleases in eukaryotic cells. Further, she noted the U.C.’s own inventors (specifically, Dr. Doudna) observed that there were engineering problems with applying CRISPR-Cas9 to eukaryotic cells, and that there was a “huge bottleneck” in modifying genetic structures in animals and humans (i.e., eukaryotes). The testimony was further supported by the prior art which did not assess reasonable expectation of success of CRISPR-Cas9 in eukaryotic systems. Therefore, Judge Moore noted there was “substantial evidence” to support the finding that a POSITA would have no reasonable expectation of success with CRISPR-Cas9 in eukaryotic cells. She further concluded her discussion on this topic:
We are [] an appellate body. We do not reweight the evidence. It is not our role to ask whether substantial evidence supports fact findings not made by the PTAB, but instead whether such evidence supports the findings that were in fact made. Here, we conclude that it does.5
As to the specific instructions:
In this case, the Board found there would not have been specific instructions in the art as to CRISPR-Cas9 that would have given one of ordinary skill in the art a reasonable expectation of success, and it was “persuaded that the failure demonstrated with other systems would have indicated the lack of a reasonable expectation of success.”6
As to the U.C.’s argument that the PTAB disregarded its argument of simultaneous invention, Judge Moore wrote:
Simultaneous invention may serve as evidence of obviousness when considered in light of all of the circumstances … . First, it is evidence of the level of the skill in the art. Second, it constitutes objective evidence that persons of ordinary skill in the art understood the problem and a solution to that problem. Inherent in the existence of interference practice is the principle that evidence of simultaneous invention cannot alone show obviousness, otherwise any claims involved in an interference would be unpatentable for obviousness.7
Judge Moore observed that the U.C.’s argument that its article by the inventors in August 2012, which was then used by six different groups’ research into CRISPR-Cas9 in eukaryotic cells was evidence of a motivation to combine the prior art. However, while there may have been a motivation to combine, she continued, that this does not mean there necessarily was a reasonable expectation of success prior to completion of the experimentation to invoke a simultaneous invention. In other words, the evidence proffered by the U.C. went to a different analysis under §103.
Therefore, the panel affirmed the PTAB’s finding of no interference-in-fact.
In the bigger picture, the Fed Circuit ruling allows the U.C.’s ‘859 application to continue prosecution and probable issuance, and Broad’s ‘359 patent remains valid. It further means with two different areas in the gene editing space, with some significant overlap, any potential licensee would need to obtain a license from both the U.C. and Broad for use of the CRISPR-Cas9 methods. Given the amount of investment and licensing opportunities in this groundbreaking patent landscape, the U.C. is probably going to petition for rehearing en banc with the Fed Circuit, or even petition for writ of certiorari with the U.S. Supreme Court in order to secure full ownership in the CRISPR-Cas9 space. There will certainly be much more litigation and we will continue to follow it on this blog.
Footnotes
-
___F.3d___ (Fed. Cir. 2018) (slip op.) (“Broad” decision), aff’g The Broad Inst., Inc. v. The Regents of the Univ. of Calif., Pat. Interfer. No. 106,048 (P.T.A.B. Feb. 15, 2017) (Paper No. 892-93). ↩
-
See, e.g., Theodore L. Roth, et al., Reprogramming human T cell function and specificity with non-viral genome targeting, Nature 559:405-409 (July 11, 2018). ↩
-
See, e.g., Dimitros Vlachakis, On novel anti-viral HIV gene editing platforms, J. Gen. DNA Res. 1(1):1000e101 (July 31, 2017). ↩
-
See Martin Jinek, Krysztof Chylinski, Ines Fonfara, Michael Hauer, Jennifer A. Doudna, & Emmanuelle Charpentier, A programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity, Science 337(6096):816-821 (Aug. 17, 2012), DOI: 10.1126/science.1225829. ↩
-
See Broad, supra (slip op. at 12). ↩
-
Id. (slip op. at 13). ↩
-
Id. (slip op. at 14-15). ↩

Brent T. Yonehara
Founder & Patent Attorney
Founder Brent Yonehara brings over 20 years of strategic intellectual property experience to every client engagement. His distinguished career spans AmLaw 100 firms, specialized boutique I.P. practices, cutting-edge technology companies, and leading research universities.
More About Brent